March 2022
BREAKING NEWS
On Feb 15th, 2022, Getein One Step Test for SARS-CoV-2 Antigen (Colloidal Gold) was approved by Coronavirus Test Device Approvals (CTDA) of Department of Health and Social Care (DHSC) in the UK.
This is the FIRST COVID-19 antigen self-test kit (nasal swab) approved by CTDA, which means that Getein One Step Test for SARS-CoV-2 Antigen (Colloidal Gold) is now permitted to enter the UK market.
*MHRA is an executive government agency under the Ministry of Health of the UK, which ensures the safety and effectiveness of medicines and medical devices.
Link: https://www.gov.uk/government/publications/covid-19-test-validation-approved-products

WHO Recommendation
According to WHO, as a diagnostic method of SARS-CoV-2 infection, Ag-RDT (Antigen-detecting Rapid Diagnostic Test) plays an important role in patient management, public health prevention and control, and COVID-19 monitoring during this pandemic, especially in areas where clinical diagnosis, treatment and prevention and control of COVID-19 are difficult due to lack of nucleic acid detection ability. In addition, Ag-RDT can offer a faster and less expensive way to diagnose active SARS-CoV-2 infection than nucleic acid amplification tests (NAATs)
.
Getein Antigen Self-Test
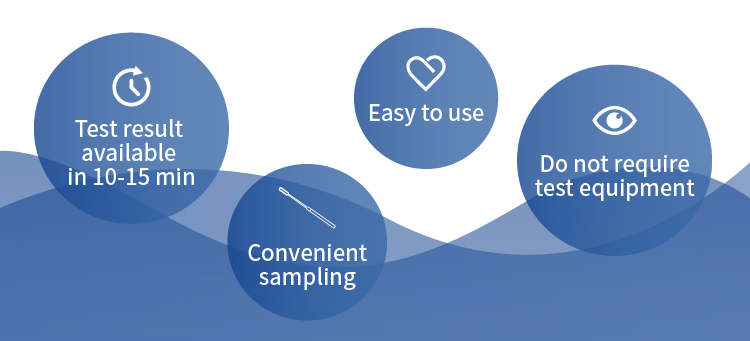
One Step Test for SARS-CoV-2 Antigen (Colloidal Gold) is intended for the qualitative detection of SARS-CoV-2 antigens in human nasal swab samples. This test is suitable for medical laypersons as a self-test at home or at work.
The test kit is easy and fast to operate without detection equipment. Users can collect nasal swabs by themselves at home, and the detection results can be obtained in only 10-15 minutes, which is very convenient for individuals and families to have a quick detection.

Multiple Certifications, Quality Guaranteed
In addition to the UK CTDA, Getein One Step Test for SARS-CoV-2 Antigen (Colloidal Gold) is also CE marked and listed in the EU common list. Besides, the test kit has also passed the PEI evaluation and the phase 3a validation of British DHSC. What’s more, Getein One Step Test for SARS-CoV-2 Antigen (Colloidal Gold) is validated and approved by Thailand FDA, Malaysia MDA and Philippines FDA.
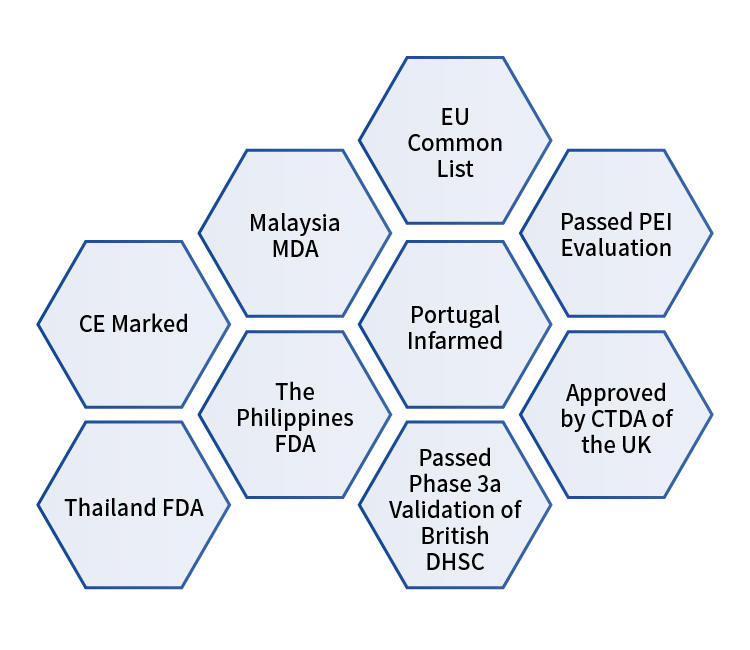
These certificates and validations provide strong proof for the high quality and good accuracy of the test kit. Just as a performance evaluation conducted by Centralne Laboratorium Kliniczine Centrum Kliniczne (Commissioned by Getein Biotech, Inc.), the diagnostic sensitivity is 96.43% and the diagnostic specificity is 100%
During this long fight with COVID-19, Getein Biotech Inc. continuously focuses on the pandemic situation and contributes itself to the prevention and control of this public crisis, so as to promptly support the resumption of the global work & production as well as the early return of people’s normal life.
For any needs, please email us via sales@getein.com.cn

Open WeChat and Scan the QR Code. Stay Tuned with Us.